Research
Structure-Function Relationships of Protein Mimics and Peptide Natural Products
We seek to mimic and improve upon nature through innovations in molecular design and synthetic approaches.
Our lab uses the tools of organic chemistry to...
Our lab uses the tools of organic chemistry to...
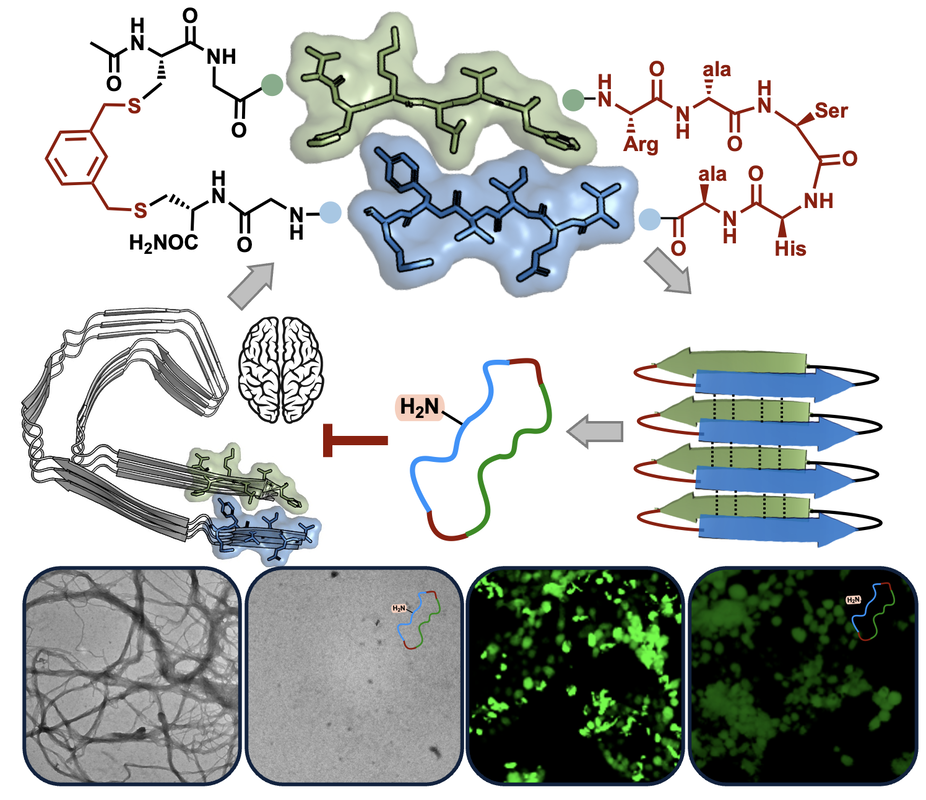
…probe neurodegenerative tau propagationAggregation of the tau protein involves conformational reorganization into parallel β-sheet structures and assembly into neurofibrillary tangles that are characteristic of a wide array of diseases. Pathological conformations of misfolded tau propagate from diseased to healthy cells with high fidelity, leading to neuronal death. Targeting specific conformations of amyloid-forming proteins remains a significant challenge. We are carrying out structure-based design and chemical synthesis of tau mimetics to target disease-specific folds. We then employ biochemical and biophysical assays to evaluate these compounds for their ability to modulate tau assembly. Our goal is to develop new model systems of tau aggregation as well as potent inhibitors of fibril seeding across cells. These compounds will enable a robust examination of the conformational strain model of tau propagation in Alzheimer's and related diseases.
|
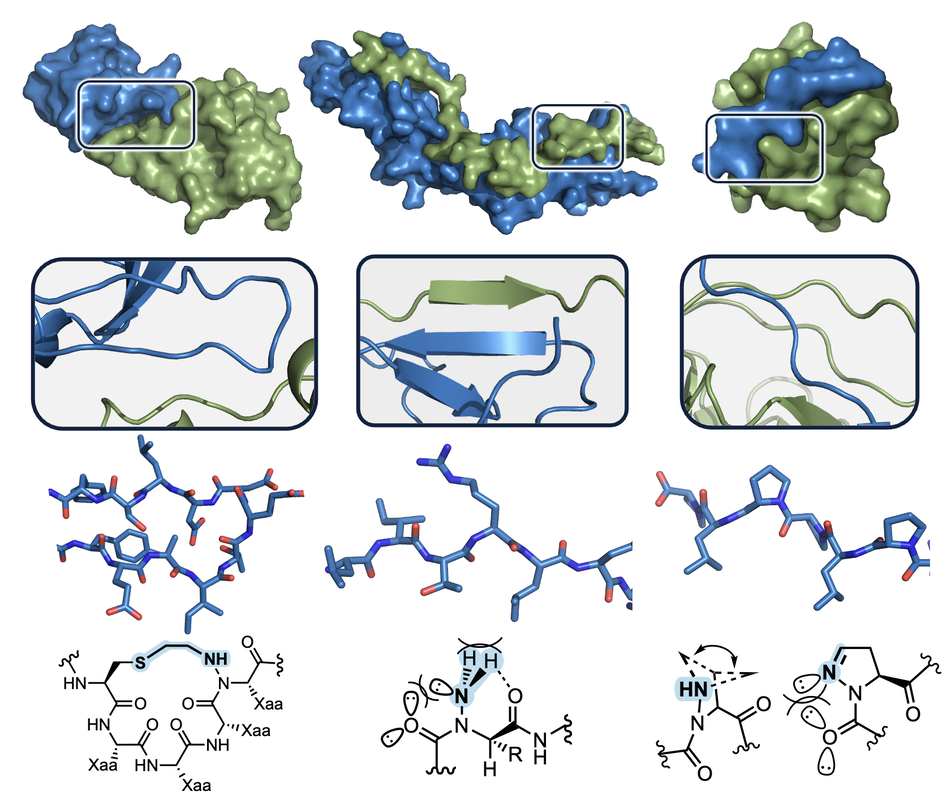
…target intractable protein-protein interactionsA myriad of biological processes are mediated by interactions between proteins. These interactions often span multiple domains, highlighting the importance of secondary and tertiary structure for biomolecular recognition and function. Although chemists have long sought to mimic protein folds through rational design, there remains a need for residue substitutions that can predictability stabilize these folds without sacrificing sidechain content. We are developing efficient synthetic routes toward soluble and stable mimics of β-sheets, polyproline II helices, α-helices, and protein loops; and in parsing the impact of discrete chemical modifications on folding. In addition to traditional covalent tethering, our designs leverage stereoelectronic effects and non-covalent interactions to influence structure. We then design hybrid or 'artificial' biomolecules with the goal of disrupting abberant protein-protein interactions in human disease.
|
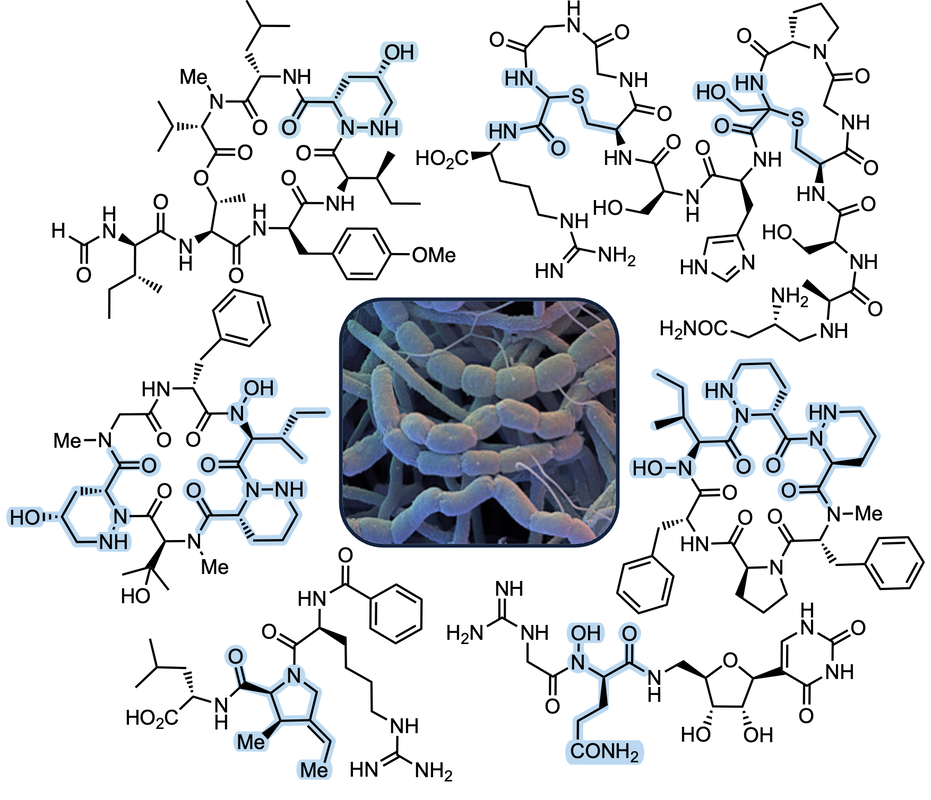
...synthesize and optimize bioactive natural productsNature remains a prolific source of biologically active drug candidates, with almost half of all FDA-approved therapeutics classified as natural product derived or inspired. Non-ribosomal peptides (NRPs) and ribosomally-synthesized/post-translationally modified peptides (RiPPs) are particularly promising classes of natural products with diverse biological activities. These structurally complex molecules are predisposed to interact with protein receptors and often feature drug-like properties not typically seen in canonical polypeptides. We are pursuing the synthesis of several NRPs and RiPPs that harbor backbone modifications important for biological activity. Total synthesis provides a platform for the development of new tactics and methodologies, as well a launching point for structure-activity relationship studies. We are also leveraging synthetic access to unusual amino acids to study their effects on native peptide conformation and their utility in other drug design applications.
|
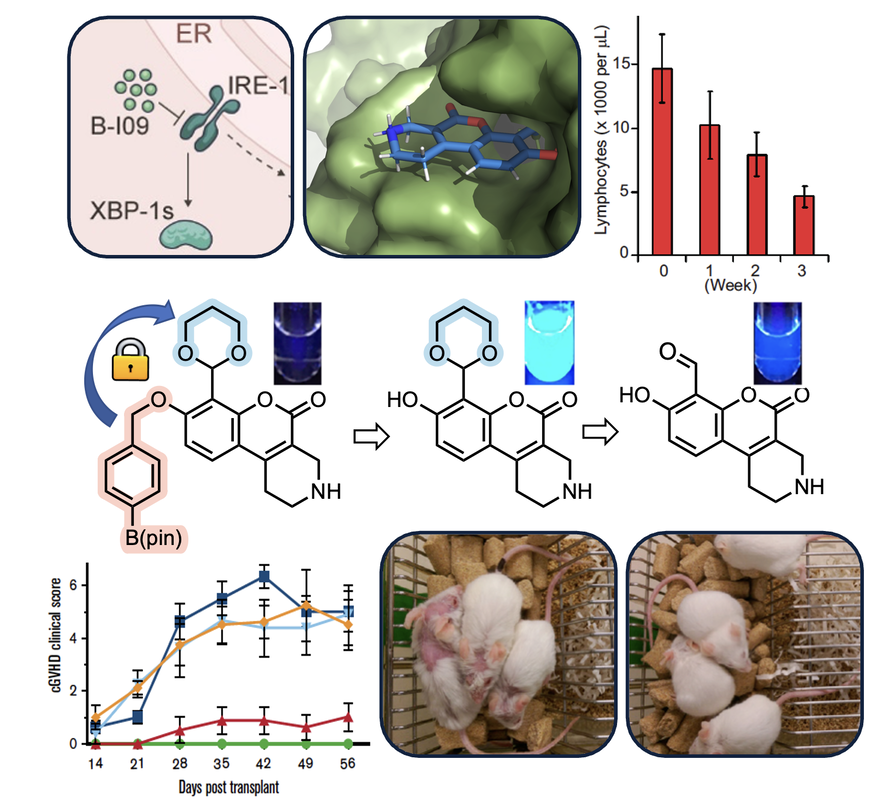
...modulate the unfolded protein response in diseaseThe unfolded protein response (UPR) is activated upon exposure to various stress stimuli and is crucial for regulating cell survival. Endoplasmic reticulum (ER) stress resulting from gene amplification and aberrant protein expression is an established hallmark of cancer. As a result, many tumors hijack this response mechanism in order to evade programmed cell death. As part of a highly collaborative effort with immunologists, cell biologists, and clinicians, our lab has developed a series of potent small molecule inhibitors of the IRE1/XBP1 signaling arm of the UPR. These compounds have helped to establish the clinical relevance of targeting IRE1 in a variety of disease models including chronic lymphocytic leukemia, c-Myc-driven cancers, and graft-versus-host disease. Current efforts are aimed at chemical optimization, advanced preclinical development, and elucidating the role of ER stress response in other diseases using tissue-specific IRE1 inhibitors.
|